Biogenesis and Characterization of Pilea-Microphylla Ferric Oxide Nanoparticles
DOI:
https://doi.org/10.5281/zenodo.15803581Keywords:
Pilea-Microphylla, Iron Nanoparticles, Green Synthesis, XRD, FE SEMAbstract
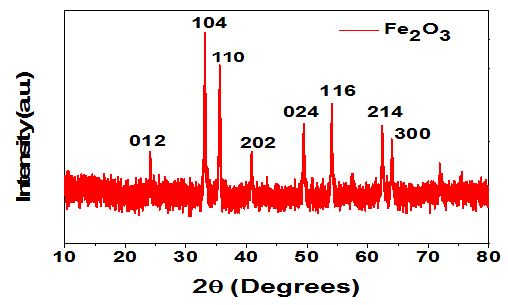
Nanotechnology involves the manipulation of materials at the nanoscale, enabling their application across fields such as environmental remediation, medicine, and engineering. However, conventional nanoparticle synthesis methods often rely on toxic reducing agents like sodium borohydride, posing environmental and health risks. This research focuses on the eco-friendly synthesis of ferric oxide nanoparticles (Fe2O3NPs) using the plant extract Pilea microphylla, a herb rich in bioactive phytochemicals such as quercetin, luteolin, and apigenin derivatives. These phytochemicals act as natural reducing and capping agents, eliminating the need for hazardous chemicals. In this study, Pilea microphylla was processed into an aqueous extract and used to synthesize Fe2O3NPs from ferric nitrate. The reaction mixture was stirred with NaOH and centrifuged to collect the nanoparticles, which were subsequently calcined at 300–400°C. The synthesized Fe2O3NPs were characterized using X-ray Diffraction (XRD) and Field Emission Scanning Electron Microscopy (FESEM) to confirm particle size and morphology. The results demonstrate that plant-based synthesis offers a scalable, cost-effective, and sustainable approach to nanoparticle production, with potential applications in wastewater treatment and environmental remediation.
Downloads
References
Saif, S., Tahir, A., & Chen, Y. (2016). Green synthesis of iron nanoparticles and their environmental applications and implications. Nanomaterials, 6(11), 209.
Huber, D. L. (2005). Synthesis, properties, and applications of iron nanoparticles.
Nasrollahzadeh, M., Atarod, M., Sajjadi, M., Sajadi, S. M., & Issaabadi, Z. (2019). An introduction to green nanotechnology. Academic Press.
Karunakaran, S., Ramanujam, S., & Gurunathan, B. (2018). Green synthesised iron and iron-based nanoparticle in environmental and biomedical application: A review. IET Nanobiotechnology, 12(8), 1003–1008.
Dhuldhaj, U. P., Shaikh, A., & Puri, J. (2023). Review on green synthesis of iron-based nanoparticles for environmental applications. Journal of Chemical Reviews, 5(1), 1–14.
Fahmy, H. M., Mohamed, F., Marzouq, M. H., & El-Din, A. B. (2018). Review of green methods of iron nanoparticles synthesis and applications. Bio Nano Science, 8, 491–503.
Greeshma, G. M., Manoj, G. S., & Murugan, K. (2017). Insight into pharmaceutical importance of bryophytes. Knowledge Resource Journal, 3(1).
Bansal, P., Paul, P., Kunwar, A., Kumar, J., & Priyadarsini, I. (2012). Radioprotection by quercetin-3-O-rutinoside, a flavonoid glycoside – A cellular and mechanistic approach. Journal of Radiation Research.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Deepak Gaikwad, Prathmesh Udekar, Neelam Lohakare, Vishwanath Lad, Omkar Naik, Sahil Patil

This work is licensed under a Creative Commons Attribution 4.0 International License.
Research Articles in 'International Journal of Engineering and Management Research' are Open Access articles published under the Creative Commons CC BY License Creative Commons Attribution 4.0 International License http://creativecommons.org/licenses/by/4.0/. This license allows you to share – copy and redistribute the material in any medium or format. Adapt – remix, transform, and build upon the material for any purpose, even commercially.









